So this article is going to dive right into omega-3 fatty acids. Oh I know what you’re thinking, but there’s really nothing funny about fatty acids. In fact, I was a little disappointed that after hours of back-breaking research (which I gathered from PubMed, Wikipedia, and a 3 page brochure I found in the parking lot) I was unable to find a single connection of the name “omega-3” to any known marvel comic super hero created by Stan Lee. Stan if you’re reading this, the world is waiting for a hero, buddy.
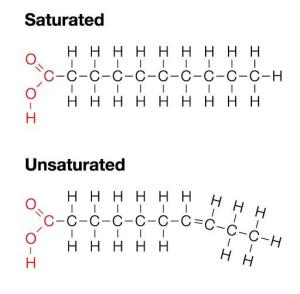
Once upon a time (called two weeks ago) I touched on saturated and unsaturated fatty acids. Feel free to glean through that article now and sing its praises on facebook and twitter if you haven’t already. I’ll wait. Let’s get to it!
So let me start by saying that omega 3 is an essential fatty acid that our body needs, and not in that Motown
“oh baby, I need your loving” kind of way, but in that “get these in your diet or else” kind of way. Anytime you see “essential” mentioned in nutrition it’s typically referring to something the body needs but can’t make effectively on its own. Babies who do not get enough omega-3 fatty acids from mom during pregnancy can develop nerve and vision problems. A deficiency in omega-3 fatty acids can lead to fatigue, a crap memory, skin like the Mojave desert, heart issues, and a bad case of the blues (mood swings, depression).
There are many types of omega 3 but the nutritional heavyweight title goes to DHA (docosohexanoic acid) and EPA (eicosapentanoic acid). These are derived almost exclusively from marine life (fish, fish oil, algae) and eggs.
There is another fancy omega 3 called ALA that we get from plant sources like flaxseed, olive oil, and walnuts but we only like it because it can be converted into DHA and EPA. Medical eggheads believe that DHA and EPA hold more nutritional punch, and as far as clinical research goes, they have a little more street credit. With that being said, if you’ve got some mouth-watering sources of ALA in your pantry don’t hesitate to tap that. Moving on…
What’s in a name?
So in reality there are about as many variations of fatty acids out there in the wild as Law & Order episodes. To distinguish between them the cool kids (scientists) describe them by the number of carbons they have and the location of the double bonds.
Remember that it’s the double bonds that make unsaturated fatty acids so kinky. The omega business is referring to the location of the first double bond that happens in the fatty acid chain, starting from the end (or omega) of the chain. So an “omega 3” fatty acid means that the first double bond is 3 carbons from the end of the fatty acid chain.
That’s it. No, don’t think any harder on that. No, look at me…let it go.
Omega 3 what have you done for me lately?
When a healthy level of omega 3 fatty acids are supplied from your diet they have the jedi ability to help lower our LDL cholesterol levels and help encourage higher, healthier HDL cholesterol levels in our blood stream. Now omega 3 has a more abstract role reducing inflammation throughout the body. Research has shown that DHA and EPA inhibit inflammation by suppressing inflammatory cytokines which are like cellular text messages that trigger cell responses.
What did inflammation ever do to us?
Well Inflammation is our body’s natural healing response when it comes to injury and infection. However, sometimes, not unlike a good friend (you know who you are), it can get a little out of hand. From an immunity stand point inflammation increases the fluid volume in the vessels and make blood vessel walls accessible for white cells to pass through in order to migrate to the source of an infection and kick ass. It also involves increased plasma flow allowing crucial proteins open access. This typically causes swelling, redness, heat, and in many cases, pain and discomfort. Fluid increases around the tissues and those tissues are surrounded by nerves usually. So as your tissues swell it pushes on the nerves and causes pain. This like many natural responses requires checks and balances. There are an [EXPLETIVE] ton of conditions involving chronic inflammation, a classic example being rheumatoid arthritis that tends to involve the small joints of the body.
So when an infection or injury is resolved you would probably like the inflammation to mellow out. Some researchers believe that part of our body’s ability to keep inflammation in check is largely associated with a balance of omega 3 and omega 6 fatty acids. By “balance” of course they mean get more Omega 3 into the diet. I personally hate this kind of health debate with the white hot intensity of a thousand suns. Both omega 3 and 6 are essential to our diet and another group of researchers in the same FRIGGIN room will say that having to balance the two is shenanigans (technical term). So my advice is make like the Eskimos (Inuit to be polite) and just eat your oily fish! If you’re on the fence about any of it just stick to eating a diet lower in saturated fat and cholesterol which will help reduce your chances of heart disease no matter which way your omega door swings.
I hope this guides you in the right direction. Stay curious, stay classy, and never stop learning my friends.